 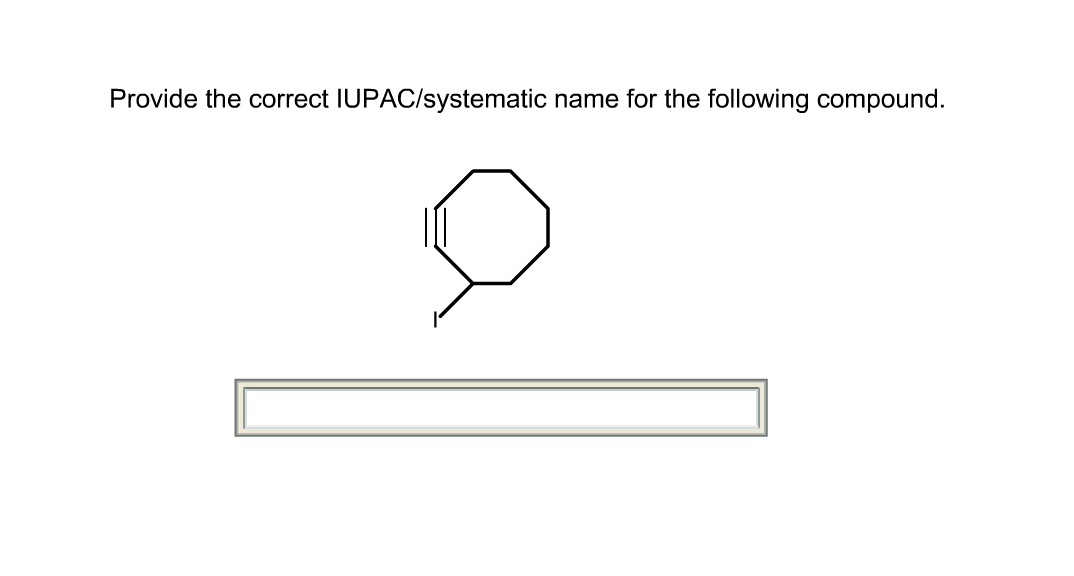
Notice again that the ethoxy group has no priority over the methyl and bromide, and they are all added as substituents in alphabetical order. With some practice they will become part of your functional vocabulary. Now, considering this, let’s name the following ether with alkyl and halide substituents: The OR group, on the other hand, has no priority and is added as a prefix in alphabetical order. The OH group has the highest priority and the parent chain is altered from “ane” to “ol”. This means that, just like alkyl groups and halides, they are only treated as substituents and therefore, they do not change the suffix of the parent chain.įor example, let’s compare the effects of the OH and OR groups on naming structurally similar compounds: The number of free calls to this function is limited. Please read the instructions below each input. It is important to mention that alkoxy groups do not rank in the priority chart of the functional groups. Use this tool to either convert drawn chemical structures into IUPAC names or to create the chemical structure from the written IUPAC name. The parent chain is determined just like we always do, based on the longest carbon chain. The idea here is to treat one of the alkoxy (alkyl with the oxygen) groups as a substituent connected to a parent chain. 
The systematic nomenclature is used for ethers with complex substituents. If the groups are identical – symmetrical ethers, the prefix “di” is added. different alkyl groups are bridged with the oxygen. The molecules above are examples of unsymmetrical ethers i.e. To do this, we first identify the alkyl groups and arrange them in alphabetical order followed by the word “ether”. The common names are used for ethers with simple alkyl groups. 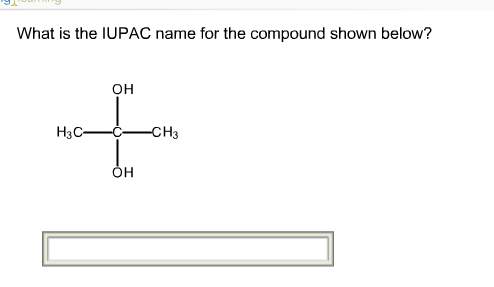
Ethers are named by both common and systematic nomenclature of the IUPAC rules. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
